The BioHealth Innovation Challenge welcomes innovative proposals to develop and operate a biosciences incubator lab in Las Vegas. The City of Las Vegas Redevelopment Agency has compiled a variety of resources for participants to utilize as they prepare their applications. Applicants are required to adhere to all applicable provisions in federal, state, city, and county law. Please email us with any questions.
Planning & Zoning
A variety of resources are available on the City of Las Vegas’s Planning & Zoning website. Participants are encouraged to review the information available on that website. The BioHealth Innovation Challenge team has compiled key resources below.
- Building & Safety Resources
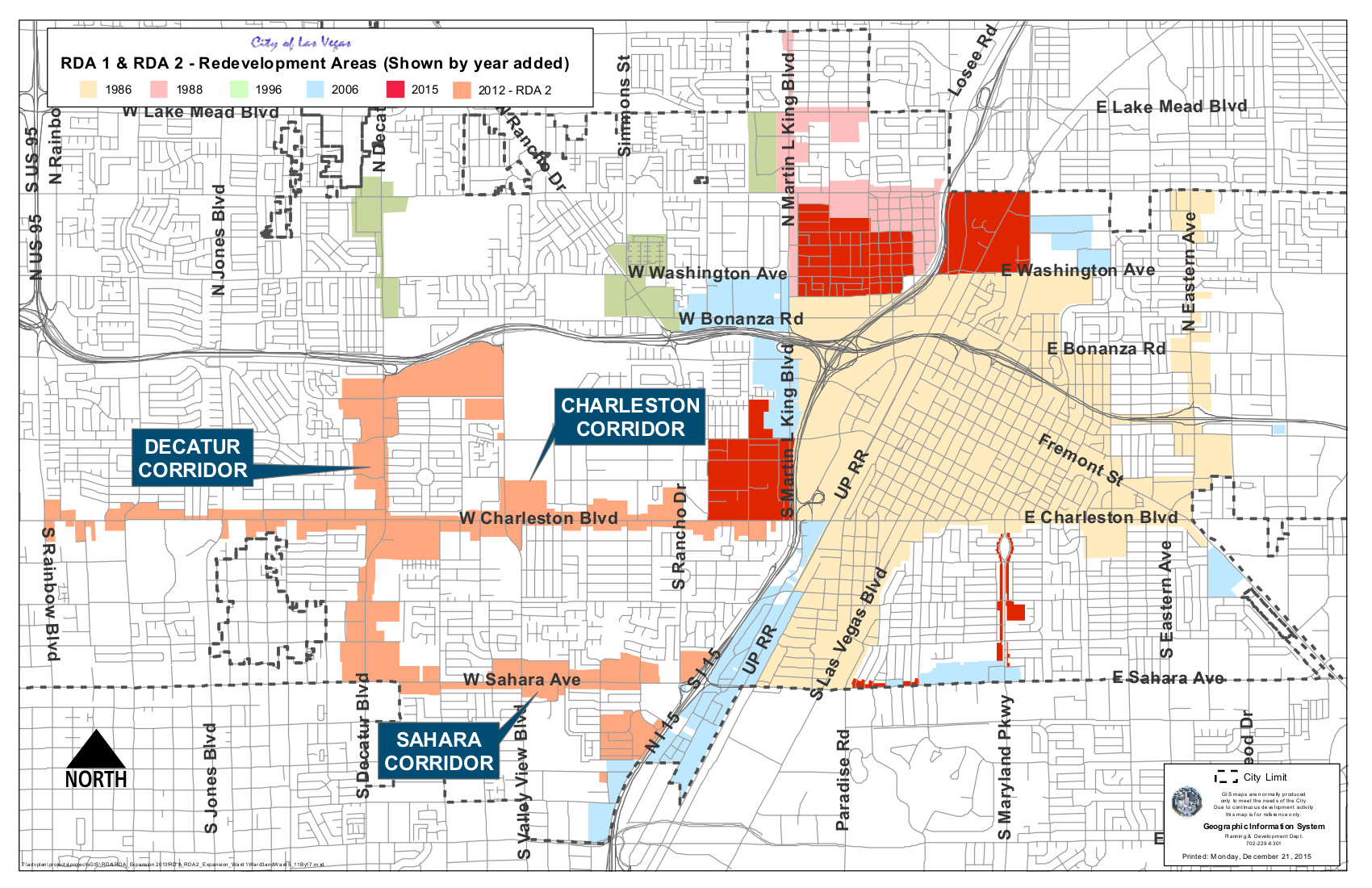
- City of Las Vegas Redevelopment Area Map
- City of Las Vegas’s Master Plan
- Department of Planning FAQ
- Form-Based Code
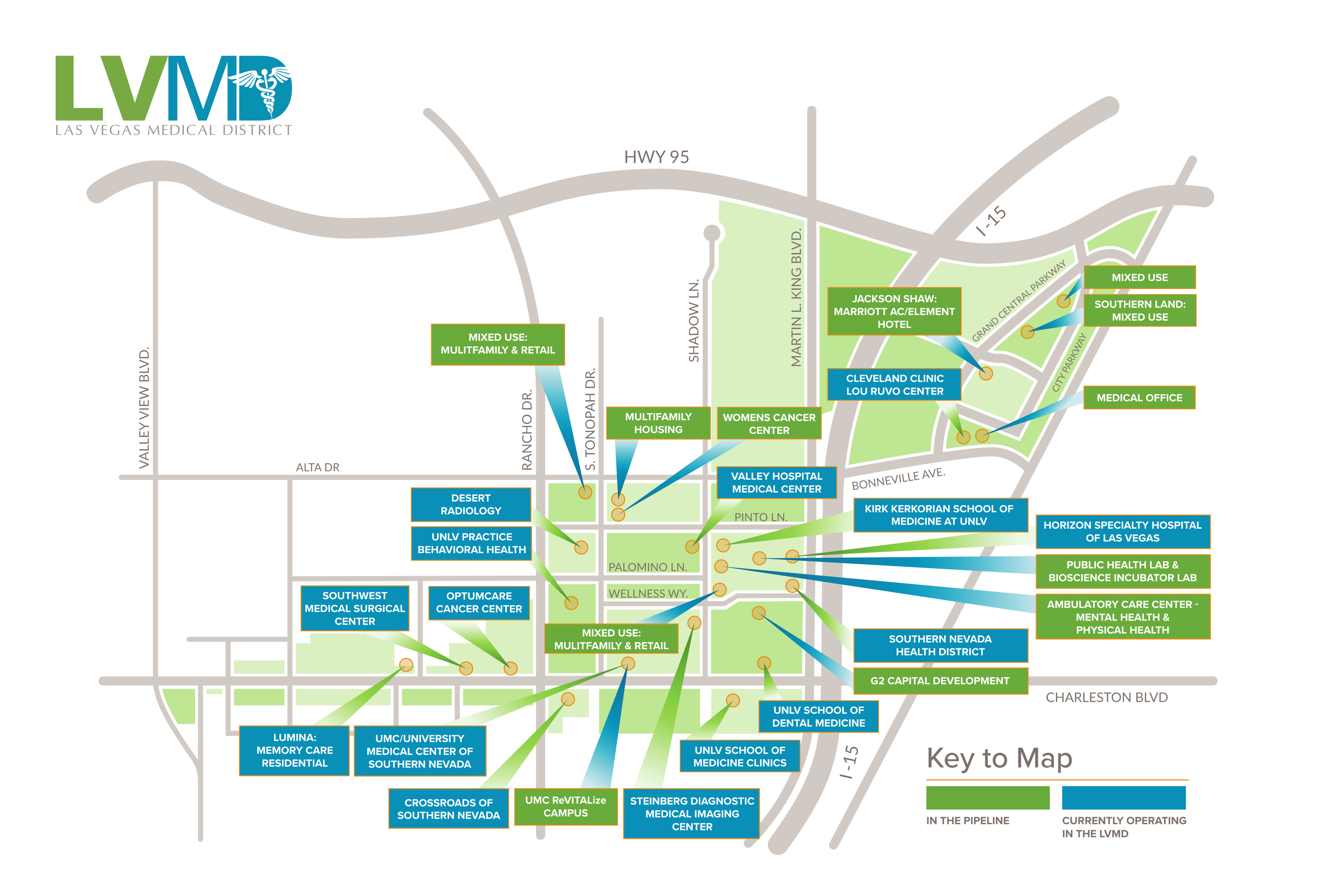
- Las Vegas Medical District Map
- Zoning Code
- Zoning Interactive Map
Forming a Business in Nevada
At the time of the award agreement’s execution, the winner must be duly organized, validly existing and in good standing under the laws of the State of Nevada and qualified to do business in the State of Nevada. Please visit the Nevada Secretary of State website to view the requirements to form a business in Nevada.
Human & Animal Subjects Research
Any applicants conducting human or animal research are required to adhere to all applicable provisions of federal, state and local law, including but not limited to, C.F.R. Title 9, Chapter I, Subchapter A, 45 C.F.R. § 46.101 et seq., the Animal Welfare Act, the Health Insurance Portability and Accountability Act (HIPAA), the U.S. Food and Drug Administration (FDA) Animal Rule, the U.S. Food and Drug Administration (FDA) Policy for the Protection of Human Subjects and other FDA regulations governing human subject protection and the conduct of clinical trials, the Public Health Service Policy on Humane Care and Use of Laboratory Animals, and Nevada Revised Statutes Chapter 574.
Regulatory Compliance
Applicants should be prepared to address compliance with any laws and regulatory frameworks that may be applicable to the project, including, but not limited to the Clinical Laboratory Improvement Amendments (CLIA), 42 C.F.R. § 493.1 et seq., the Health Insurance Portability and Accountability Act (HIPAA), OSHA Laboratory Standards, OSHA Nevada State Plan, Nevada Revised Statutes Chapter 652, Nevada Administrative Code Chapter 652, Nevada medical laboratory licensing requirements, and the City of Las Vegas Building and Safety Code.
About the Las Vegas Medical District & Our Partners
The Las Vegas Medical District brings together a variety of medical providers that surround the Kirk Kerkorian School of Medicine at UNLV, creating the region’s center of academic medicine. The district is centrally located within the Las Vegas valley and offers easy access via highways and public transportation.
Multiple medical facilities, including hospitals, the school of medicine and ancillary medical providers have created a growing and thriving medical and biosciences ecosystem within the Las Vegas Medical District. The LVMD has compiled resources and research about the District and its partners here.
There is the opportunity for potential collaboration, cooperation, and partnership between the Las Vegas Medical District, the city of Las Vegas, the University of Nevada, Las Vegas, and the winner of the BioHealth Innovation Challenge. Applicants who are interested in partnering with the University of Nevada, Las Vegas can contact David W. Hatchett, Vice President for Research, by email or at 702-895-0213.